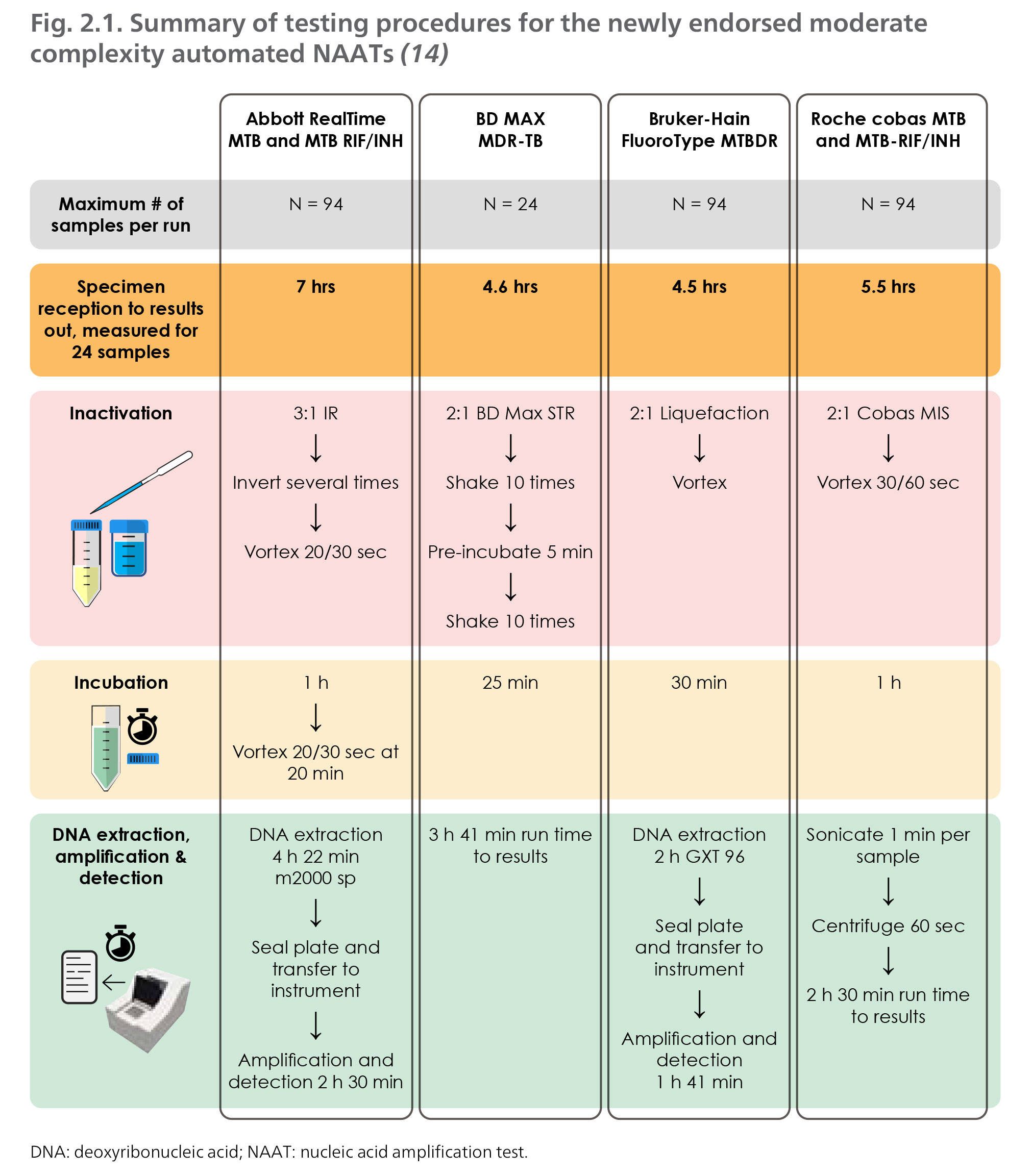
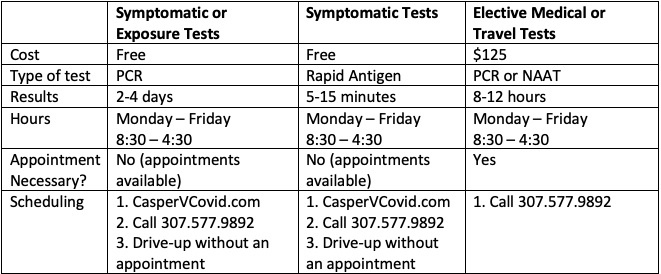
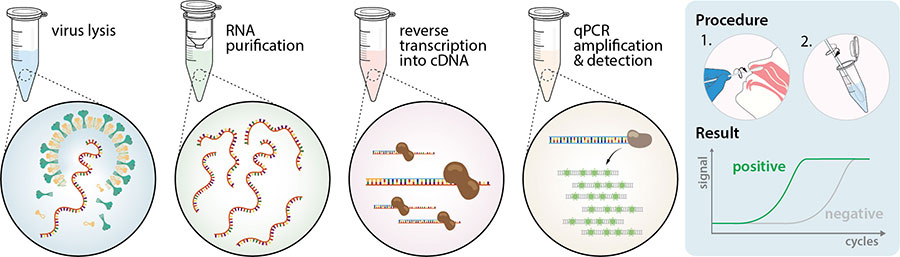
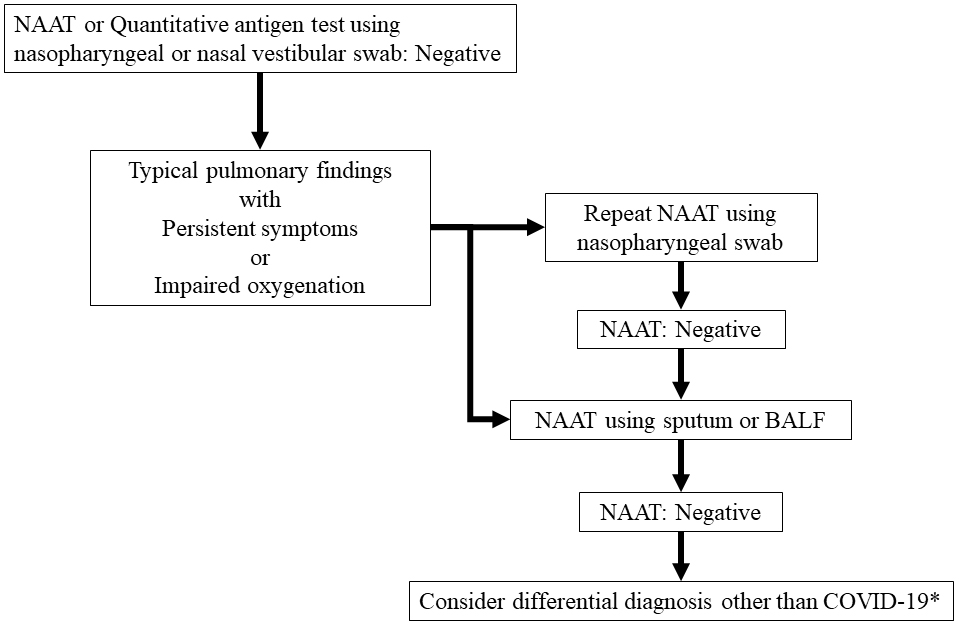
Clinical evaluation of commercial nucleic acid amplification tests in patients with suspected sepsis | BMC Infectious Diseases | Full Text
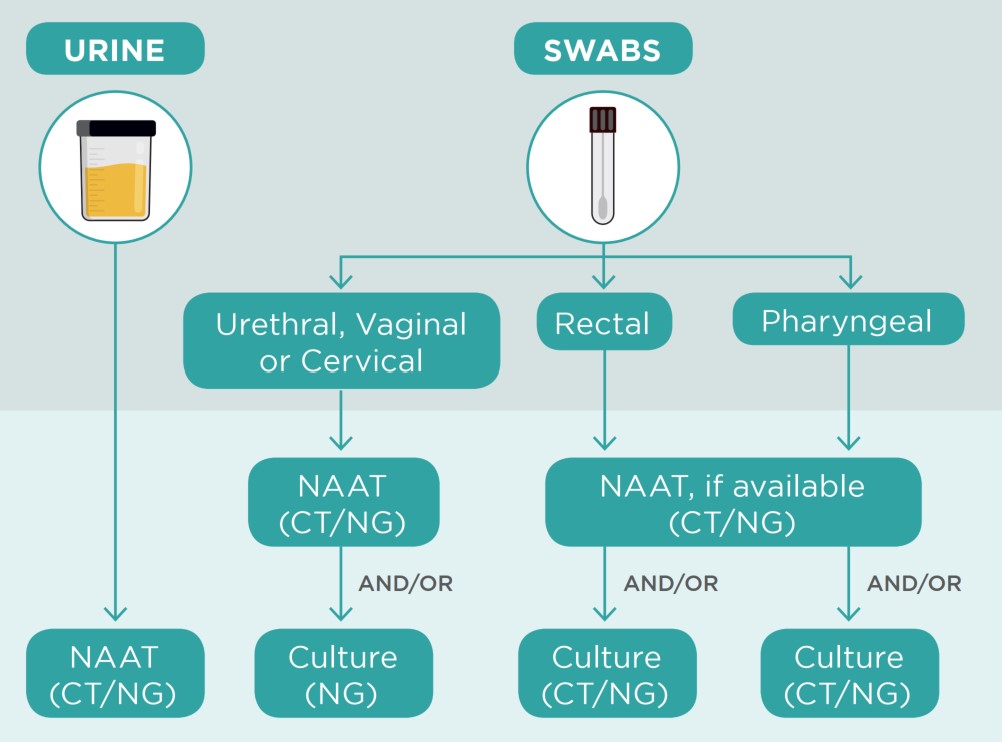
STBBI: Guides for health professionals: Summary of Recommendations for Chlamydia trachomatis (CT), Neisseria gonorrhoeae (NG) and Syphilis - Canada.ca

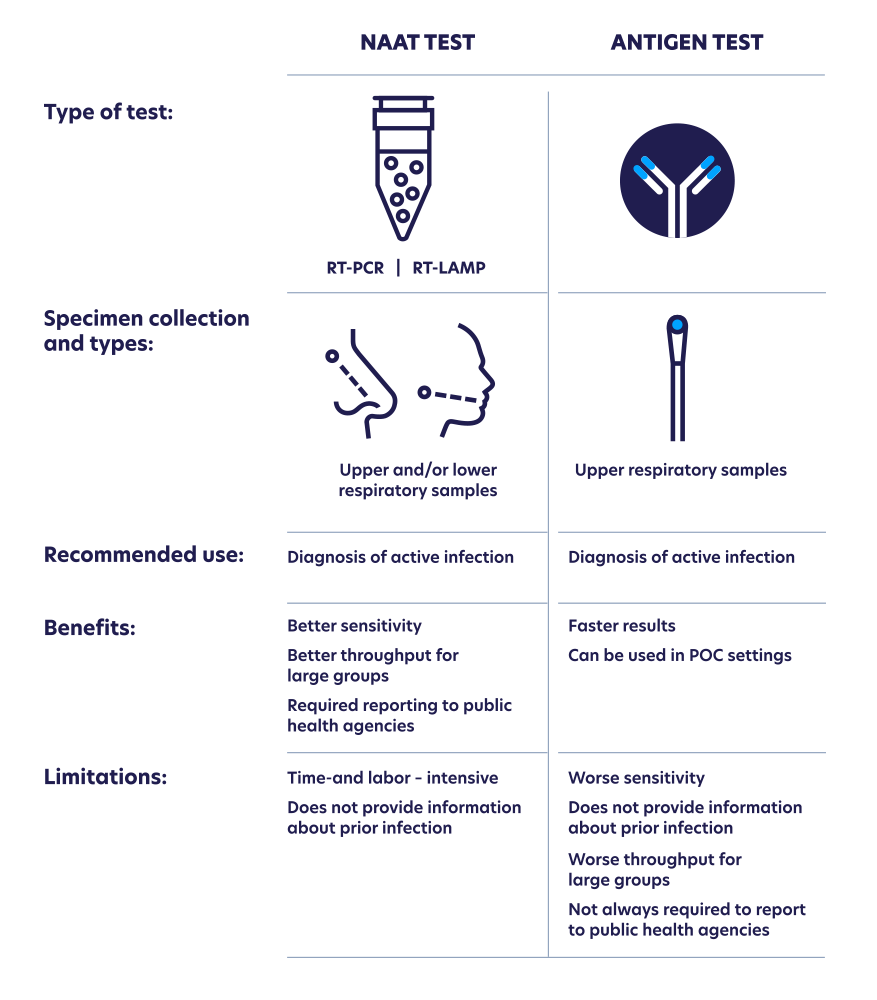
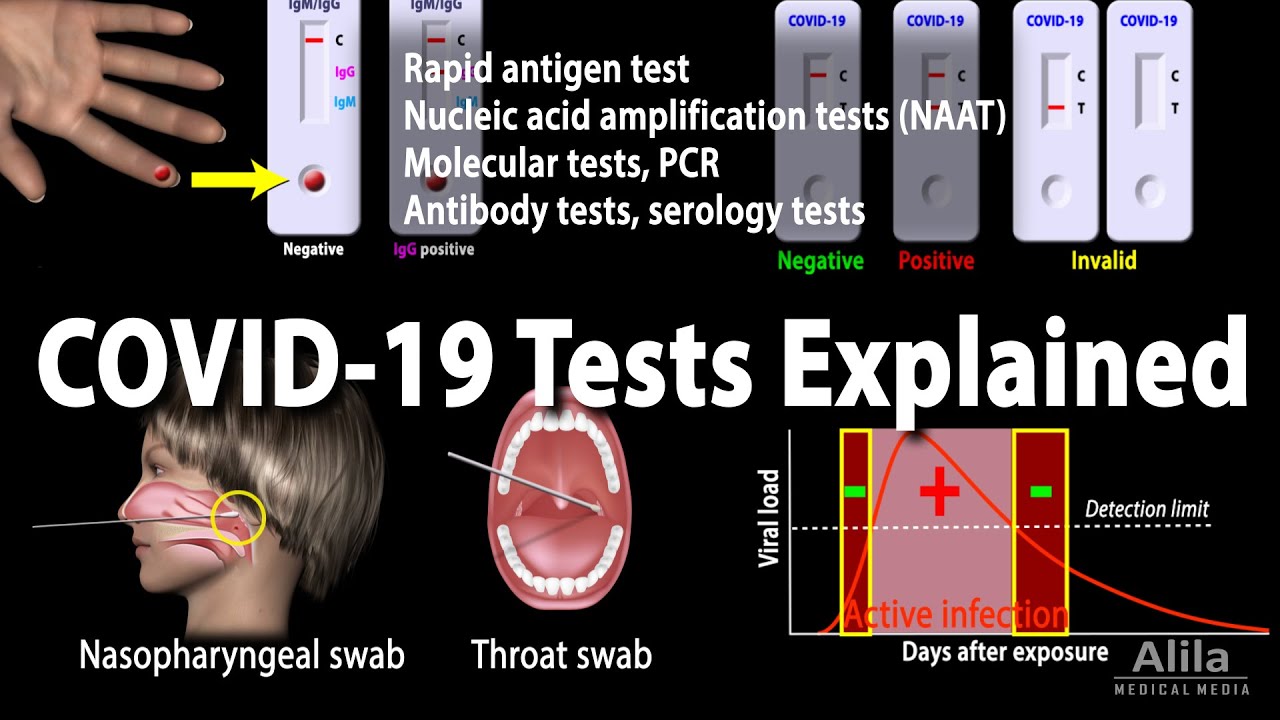
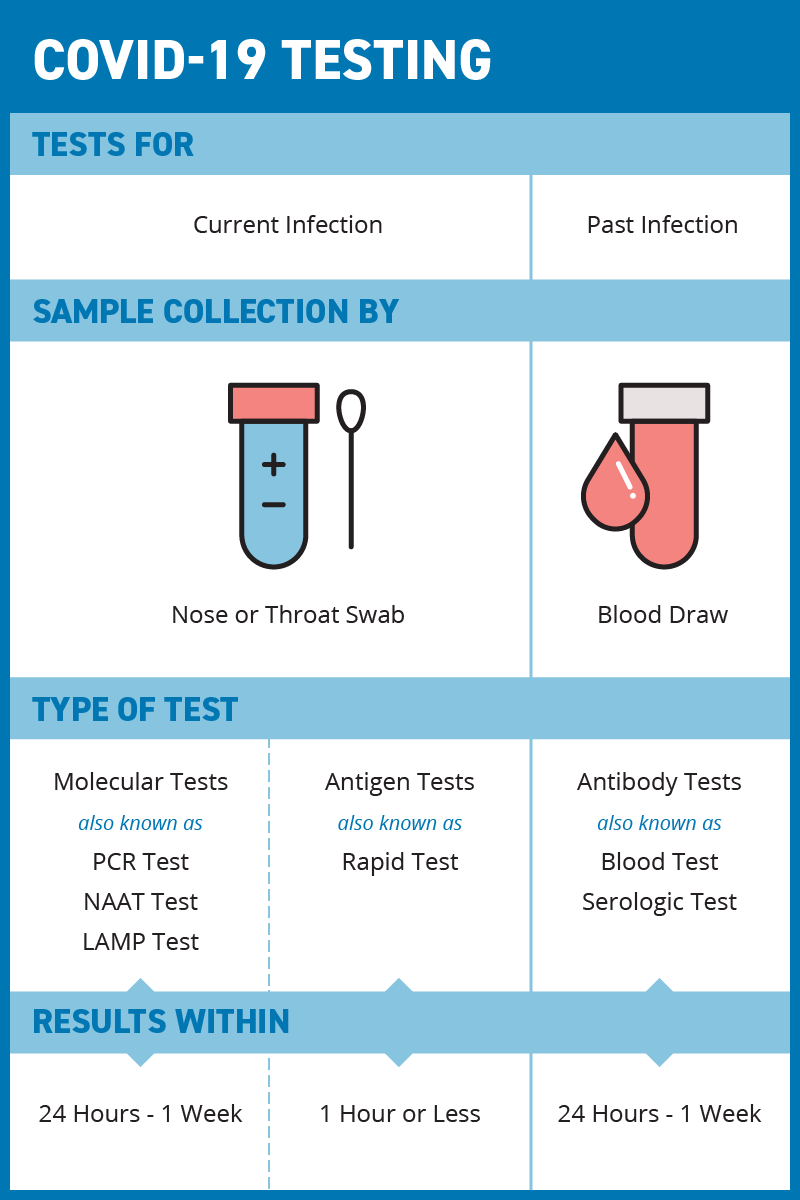
Antigen-based rapid diagnostic testing or alternatives for diagnosis of symptomatic COVID-19: A simulation-based net benefit analysis | medRxiv
Pelvic inflammatory disease risk following negative results from chlamydia nucleic acid amplification tests (NAATs) versus non-NAATs in Denmark: A retrospective cohort | PLOS Medicine

A precise review on NAATs‐based diagnostic assays for COVID‐19: A motion in fast POC molecular tests - Maleki - 2022 - European Journal of Clinical Investigation - Wiley Online Library

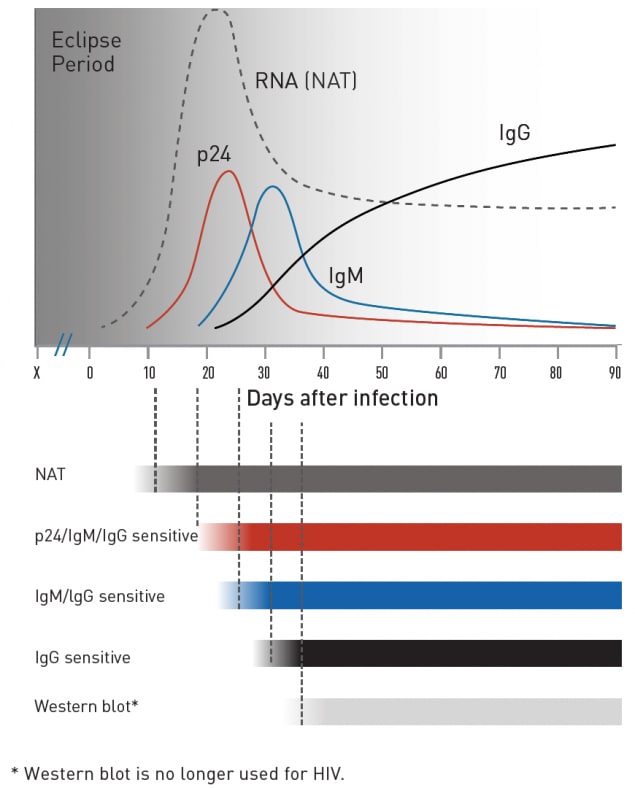
The role of nucleic acid amplification test (NAAT) in the diagnosis... | Download Scientific Diagram

A 30-Min Nucleic Acid Amplification Point-of-Care Test for Genital Chlamydia trachomatis Infection in Women: A Prospective, Multi-center Study of Diagnostic Accuracy - eBioMedicine
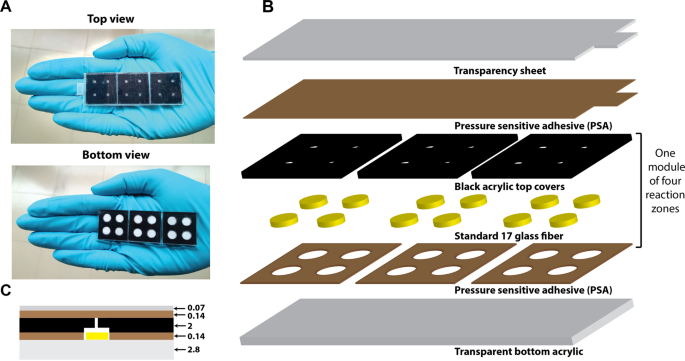
A modular paper-and-plastic device for tuberculosis nucleic acid amplification testing in limited-resource settings | Scientific Reports

American Citizen Services, U.S. Embassy Tokyo - All air travelers including U.S. citizens to the #UnitedStates are required to get a negative #COVID19 NAAT or #Antigen test within 3 days before their